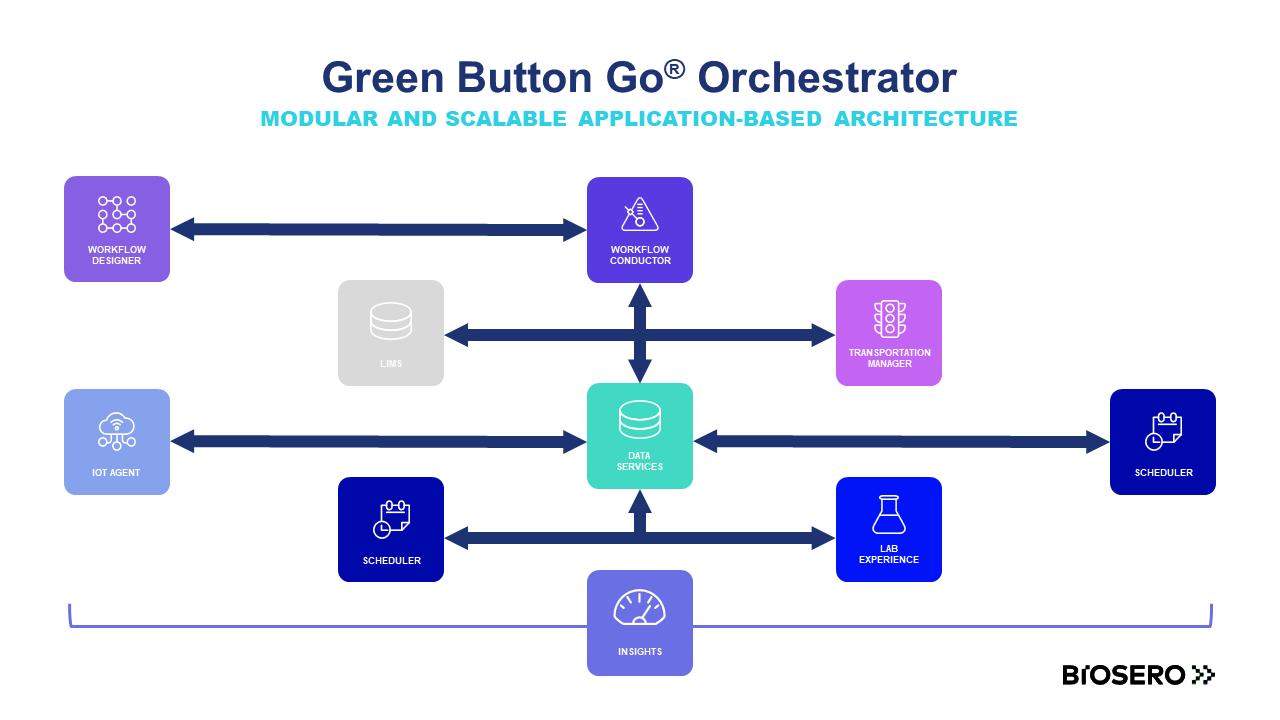
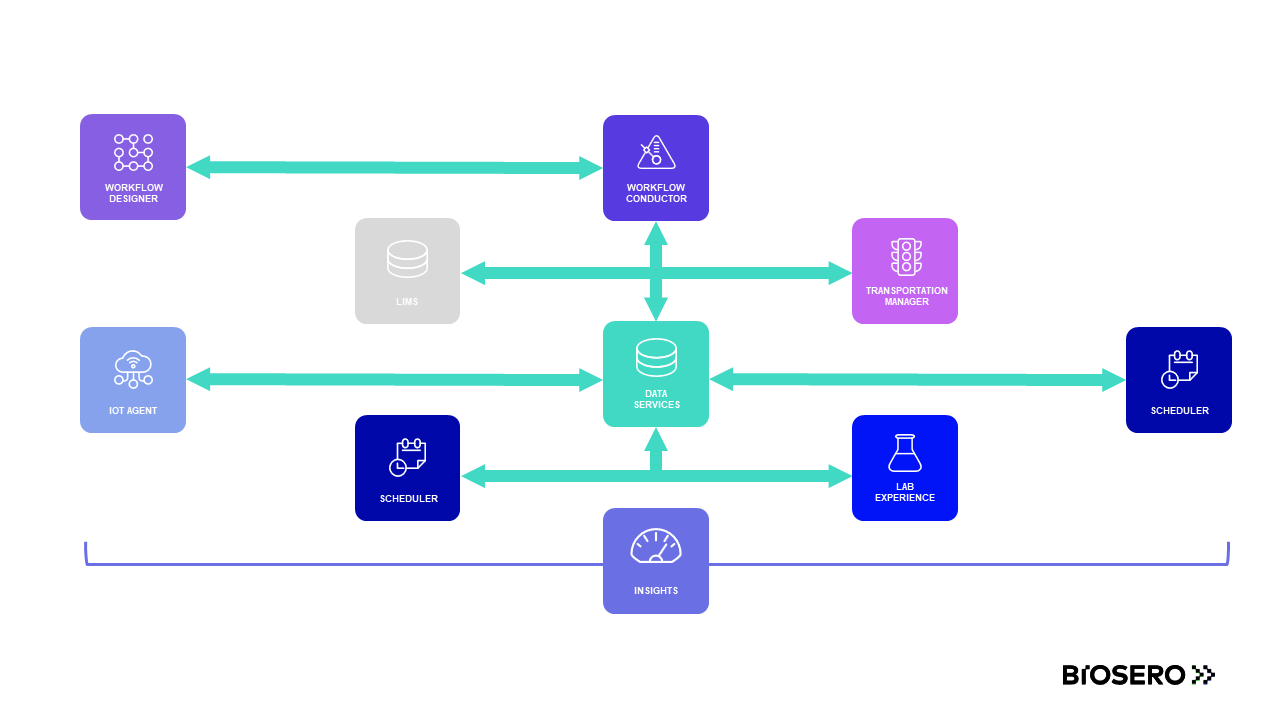
The 21CFR11 Manager extension for Green Button Go Scheduler is a seamless way to integrate tracking and validation with your laboratory automation platform. 21CFR11 Manager builds on the flexibility of GBG Scheduler by incorporating authenticity, integrity, and confidentiality of electronic records.
For regulated labs, traceability, reproducibility, and accuracy of electronic records are required to comply with the Food and Drug Administration (FDA) part 11 of Title 21 of the Code of Federal Regulations. Green Button Go Scheduler software can help get your lab into compliance.
 21CFR11 Manager
21CFR11 Manager